-
我国有色金属冶炼、硫酸生产、金属加工和电子信息等行业会产生大量成分复杂的强酸性废水。此类废水酸度极高(1%~80%),且组分复杂多变,含有铜、镍、锌等多种重金属[1-4]。受酸、有机质和盐等复杂组份的影响,常规分析方法难以对强酸性废水中的重金属进行快速准确地检测。例如,用电感耦合等离子体发射光谱法(ICP-OES)、火焰法/石墨炉-原子吸收法和电感耦合等离子体质谱法(ICP-MS)等对强酸废水中重金属分析前,为排除样品基底干扰,常需增加稀释、除酸和消解等程序。这些预处理程序不仅分析时间较长,且复杂的过程会导致分析准确性的下降 [5-11]。因此,对其中重金属的快速、准确分析是目前强酸性废水处理中的难点之一。
激光诱导击穿光谱法(laser induced breakdown spectroscopy,LIBS)是一种快速、受样品性质和其他离子干扰小的元素分析方法,可用于液体和固体样品中目标重金属的快速检测分析[12-14]。但在对液体样品进行直接分析时,还存在样品飞溅、样品表面振动以及产生的等离子体寿命短等缺陷[14]。目前,在用LIBS分析液体样品时,常常将液体样品进行固化处理。例如,沉淀富集法采用沉淀剂将液体样品中的重金属沉淀、过滤,并富集到滤膜上,以用于LIBS法检测液体样品中的重金属[15]。但在强酸条件下,有些重金属无法有效沉淀,而强酸废水中的高浓度酸会腐蚀滤膜,不利于沉淀的分离。另外,聚乙烯醇(polyvinyl alcohol,PVA) 加热成膜法也是LIBS分析液体样品时常用的固化方法[16],但因为强酸废水的强腐蚀性,受热条件下会使PVA分解,故无法实现对强酸性废水的成膜固化。因此,亟需研发LIBS分析强酸性废水时对于水样的快速有效固化方法。
一定浓度的丙烯酸和丙烯酰胺在紫外光照条件下可通过光引发剂引发聚合,长链聚合物之间形成氢键,从而使溶液变成高强度的凝胶材料[17]。受此启发,在强酸废水中添加一定浓度的丙烯酰胺和丙烯酸单体,并加入一定量的光引发剂,通过紫外光照可实现对强酸废水的快速凝胶固化。另外,已有研究表明在样品表面覆上纳米颗粒可极大增强LIBS光谱强度,提高LIBS对样品中目标元素的检测效果 [18-19]。基于此,本课题组研发了一种紫外光照促使强酸废水快速凝胶固化的方法,并采用纳米颗粒增强LIBS对固化后样品进行分析,继而定性定量分析强酸废水中的重金属,分析紫外光照凝胶固化-纳米颗粒对优化LIBS检测方法,提升该方法对强酸性废水中重金属定量分析的检出限、精确性和稳定性,为强酸性废水中重金属的定性定量检测提供新的技术支持。
全文HTML
-
重金属铜、镍和锌离子标准溶液均为1 000 mg∙L−1,购自国家有色金属及电子材料分析测试中心;丙烯酰胺和丙烯酸均为分析纯,均购于阿拉丁试剂(上海)有限公司;纳米银粉(99.5%,60~120 nm)购买于上海麦克林生化科技有限公司。将纳米银颗粒分散在丙酮中制备成浓度为10 g∙L−1的纳米银颗粒分散液。紫外灯为高压汞灯,功率为400 W,购于北京欣恒瑞光电科技有限公司。光引发剂2-羟基-4′-(2-羟乙氧基)-2-甲基苯丙酮(98.0%)购自阿拉丁试剂(上海)有限公司,实验用水为去离子水。
-
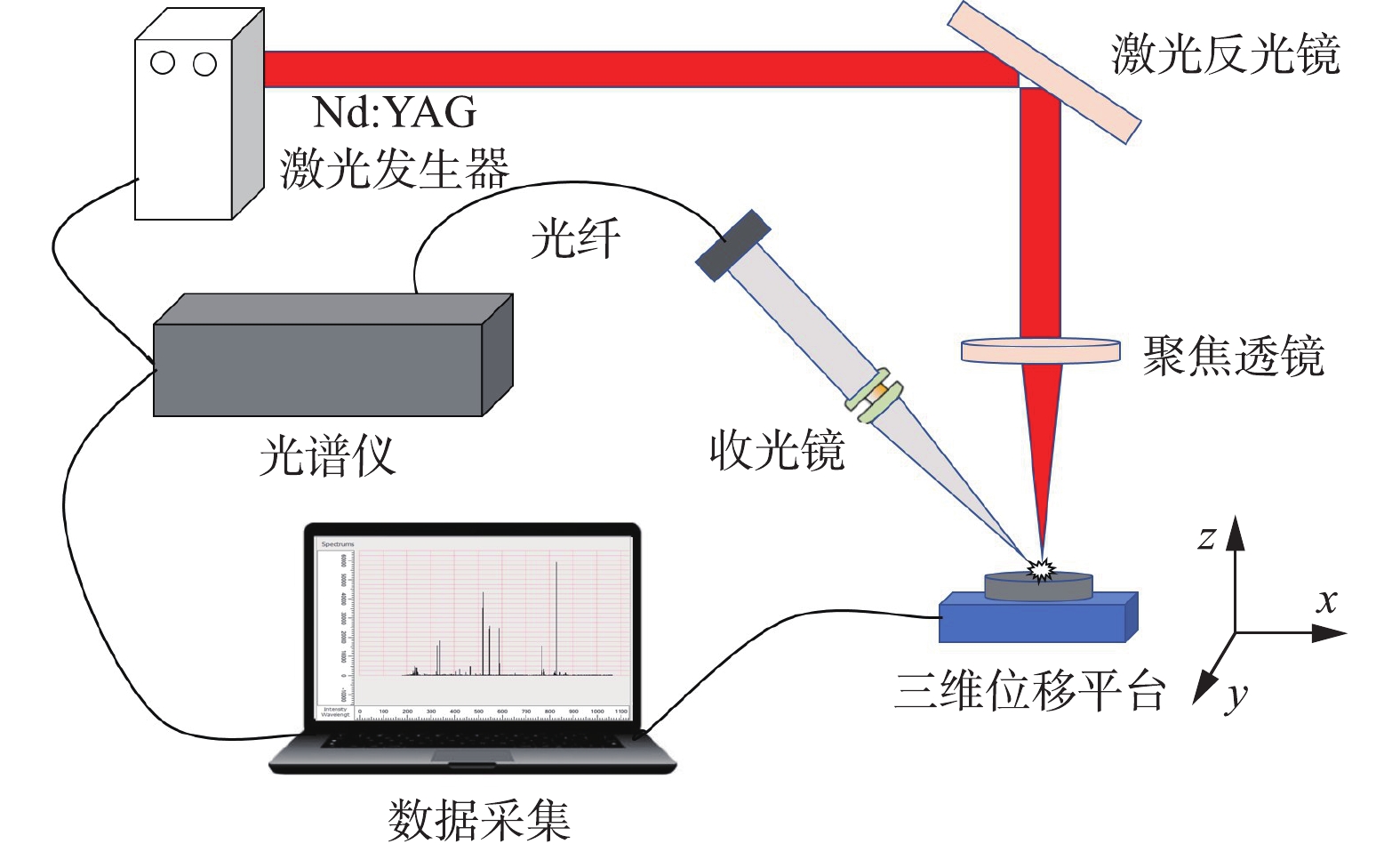
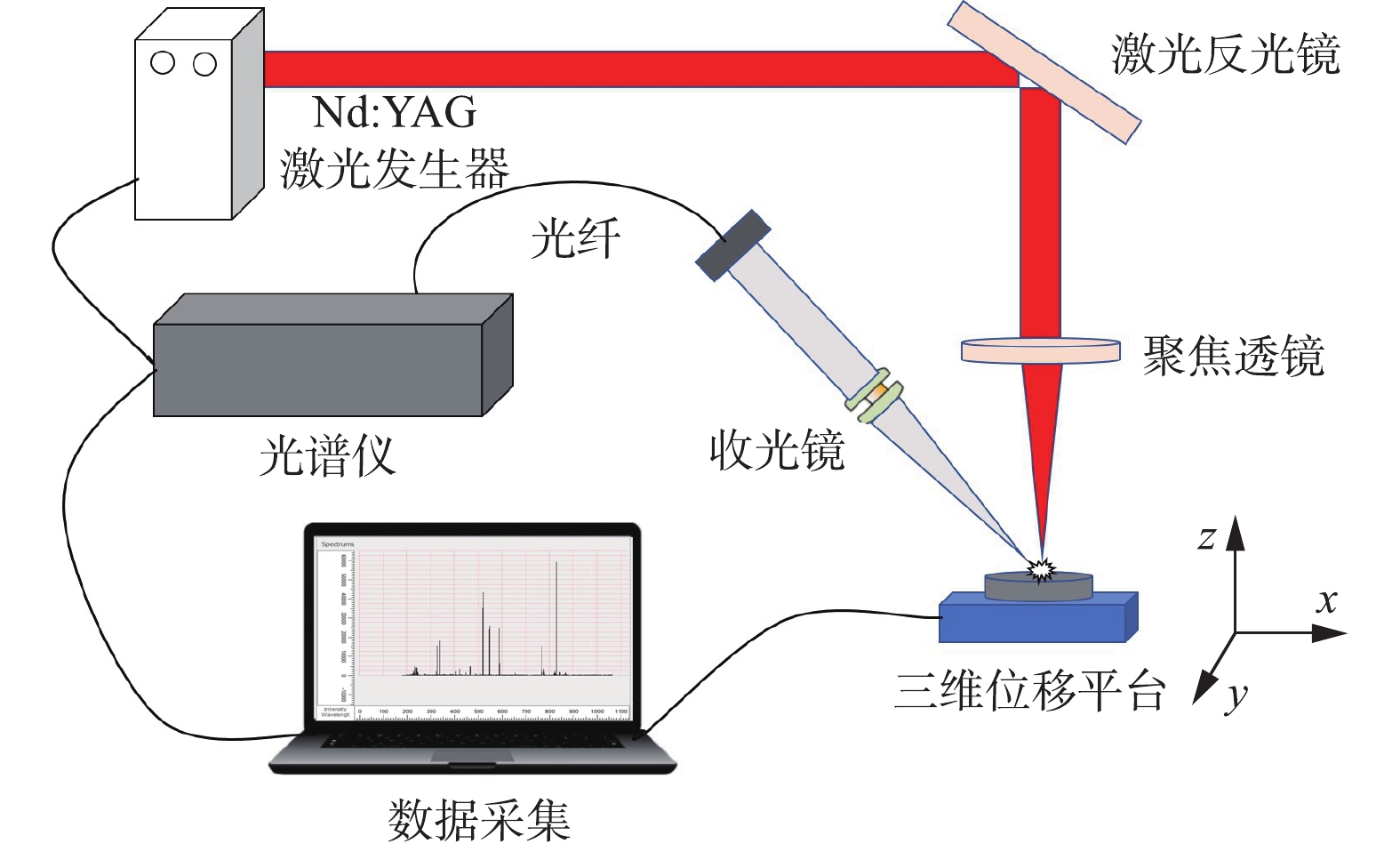
LIBS系统(Ocean optical,MX2500+)示意图见图1。激光由激光器触发产生,输出波长为1 064 nm,单脉冲最大能量为150 mJ,脉冲宽度为 6~8 ns,脉冲频率为1~15 Hz。光谱仪波长范围 180~1 037 nm,分辨率为 0.1 nm。激光脉冲与光谱仪采集的时间间隔(采集延迟时间)通过数字脉冲延时发生器控制。样品放置于配备位移控制器控制移动的三维位移平台上,操作时避免激光打在样品的同一点,以提高检测的准确性。组成该检测装置的还有:反射镜、聚焦透镜、光纤探头,光纤以及可观测样品表面的CCD显微相机。
-
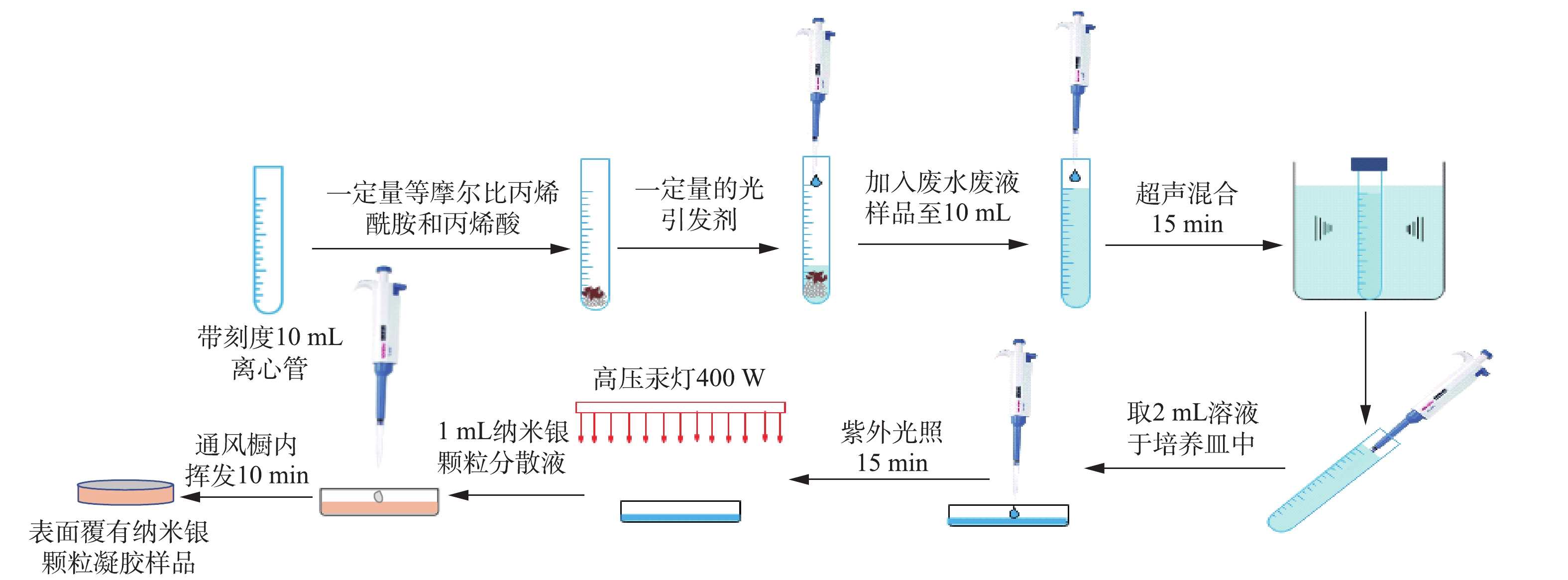
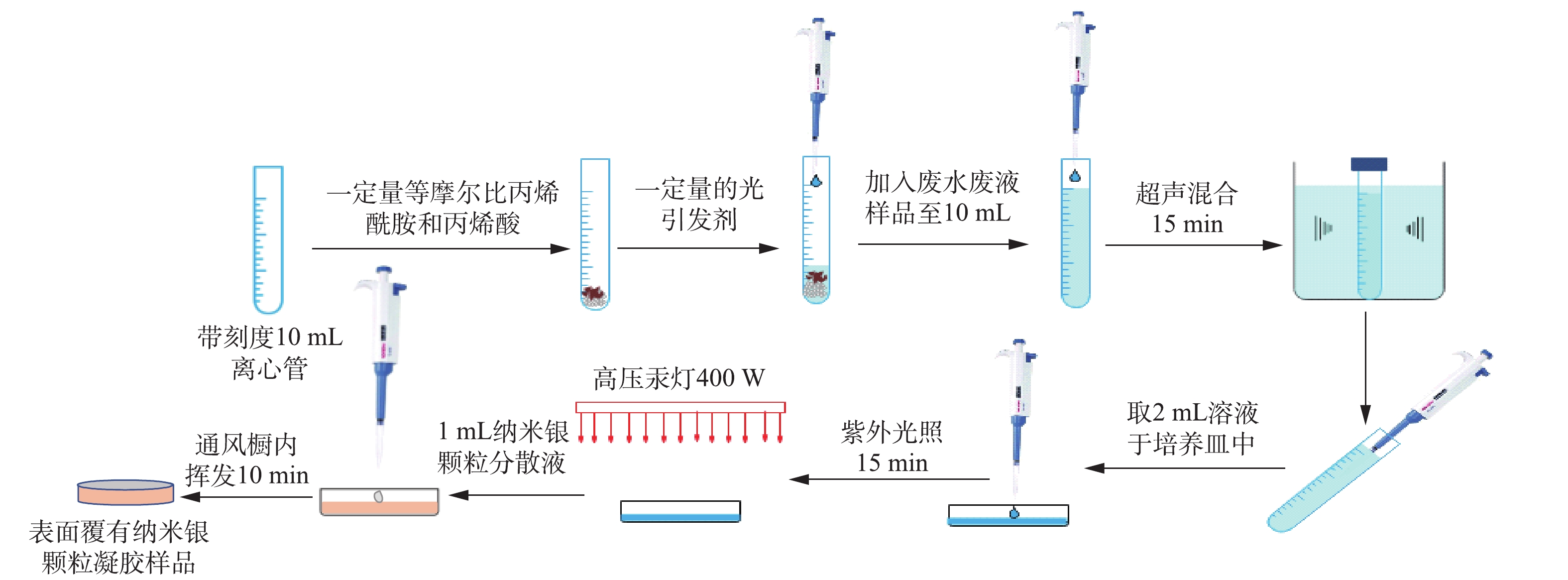
强酸性水样用浓度为1 000 mg∙L−1的铜、镍、锌离子标准溶液和浓硫酸(98%)配制。水样的详细固化流程(见图2)如下:1) 10 mL离心管中加入等物质量浓度的丙烯酰胺和丙烯酸,光引发剂(2-羟基-4′-(2-羟乙氧基)-2-甲基苯丙酮)浓度为5×103 mg∙L−1,及强酸性水样至刻度线。在超声仪中混合15 min,并取2 mL混合均匀后的溶液于直径为35 mm的玻璃培养皿中。在功率为400 W的高压汞灯下照射15 min,再冷却固化,将固化后的凝胶样品立即用于LIBS分析;2)同样按照以上步骤固化另一批凝胶样品,在样品表面滴加1 mL纳米银颗粒分散液(溶剂为丙酮,纳米银浓度为1 g·(100 mL)−1),并于通风橱中挥发10 min至干燥状态,使凝胶样品表面覆上一层纳米银颗粒,准备好的样品立即用于LIBS分析。
-
将固化后的样品放置在LIBS系统的三维位移台中,通过计算机控制三维位移台调整样品位置,使CCD显微相机能够清晰看到样品表面。打开激光控制器输出激光,设置好采集次数、激光能量和延迟时间等仪器参数,并开始数据采集。激光通过光路系统对样品表面进行轰击,激发产生等离子体,等离子体发射出光谱,利用光纤探头收集光谱信号并通过光纤将其传送到光谱仪中,最后利用LIBS光谱分析软件对采集的光谱信息进行处理分析。
1.1. 实验材料
1.2. 实验装置
1.3. 样品制备
1.4. LIBS系统对样品的检测方法
-
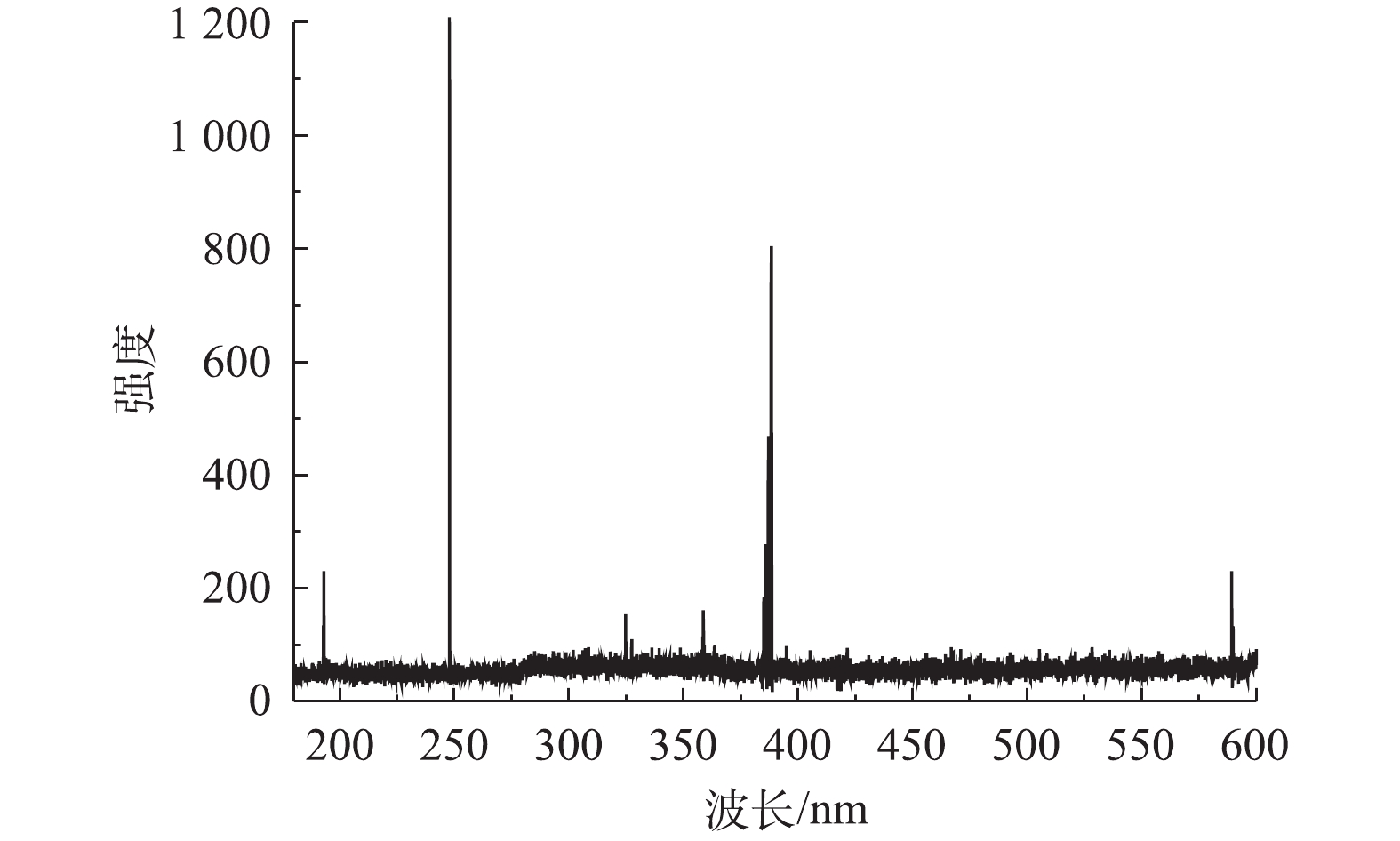
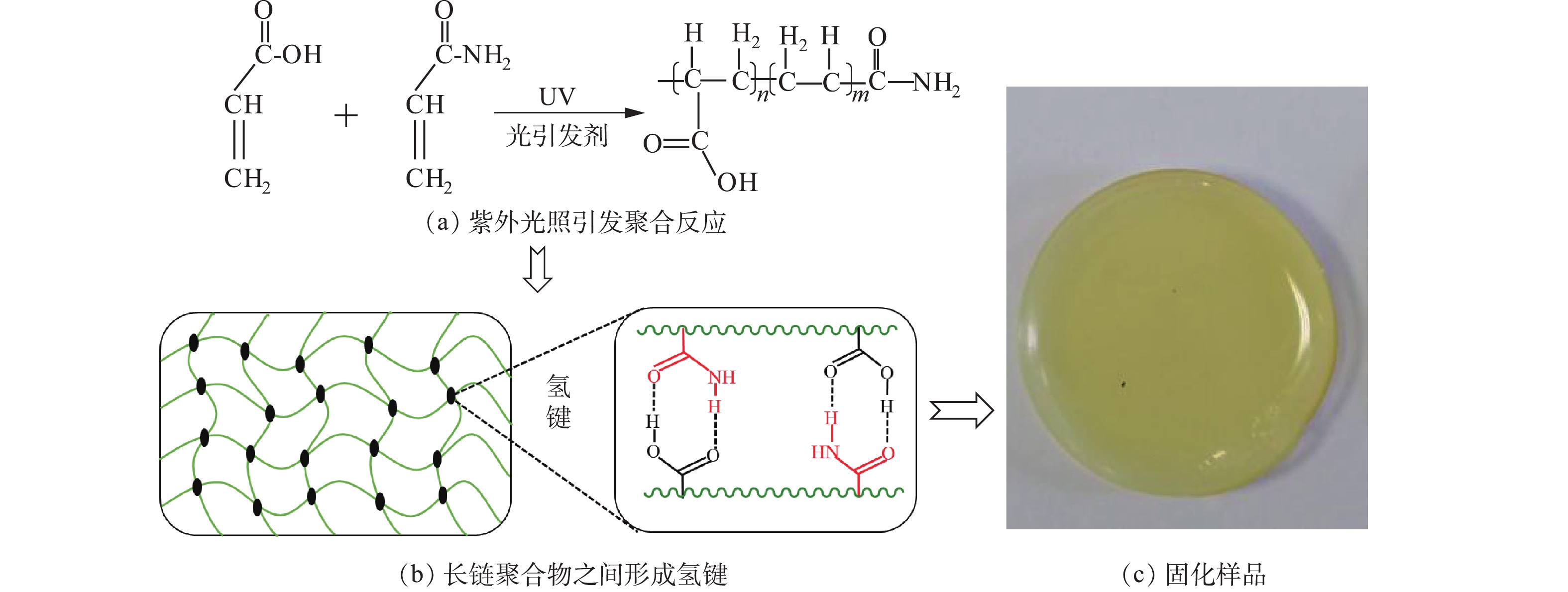
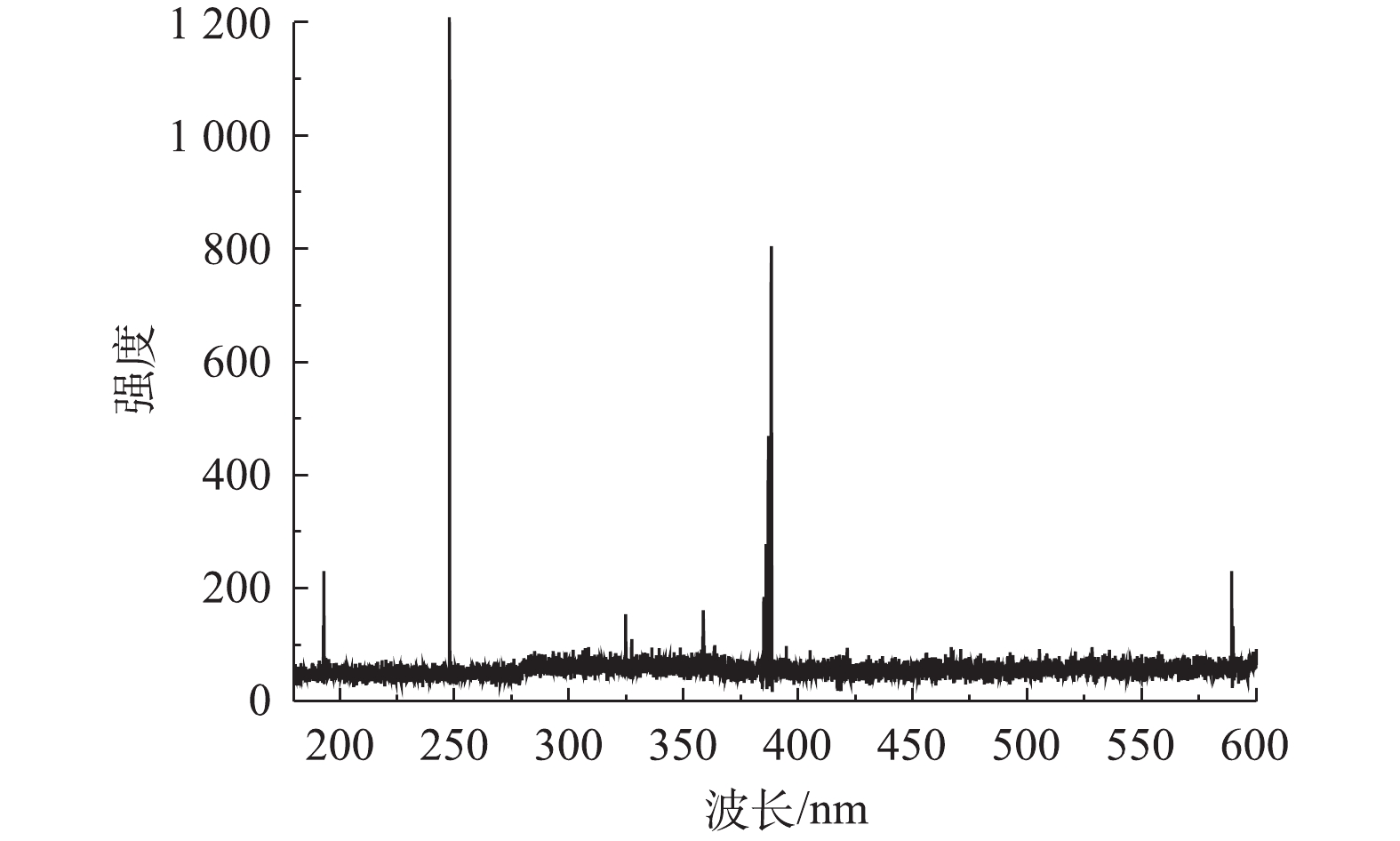
水样中Cu2+、Ni2+和Zn2+的浓度均为100 mg∙L−1,硫酸浓度为1.0×105 mg∙L−1。按照图2所示流程,向水样添加浓度为5 mol∙L−1的固化剂(等物质量浓度的丙烯酰胺和丙烯酸)和浓度为5×103 mg∙L−1的光引发剂,进行混合固化。固化剂丙烯酰胺和丙烯酸在紫外光照下,通过光引发剂引发聚合反应,从而生成长链聚合物,过程如图3(a)所示; 生成的长链聚合物支链基团之间可形成氢键,使得长链聚合物之间形成网络结构(见图(3(b)),从而使水样凝胶固化[17,20-21];固化后的样品如图3(c)所示,表明强酸性水样能够很好地被固化。将固化后的样品在激光强度为155 mJ,延迟时间为155 ns的条件下进行LIBS分析。图4表明LIBS对凝胶固化的样品进行击穿产生的光谱强度很弱,无法满足分析要求。因此,需要对凝胶固化后的样品做进一步处理,以增强LIBS对样品分析时发射光谱的强度。
-
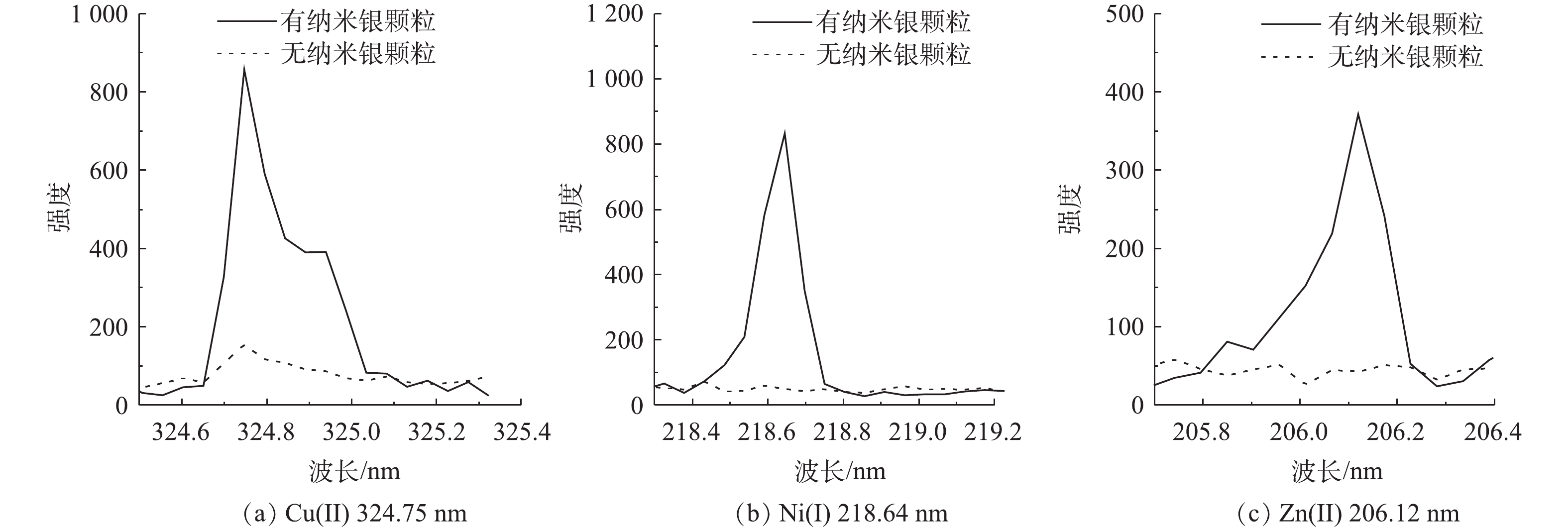
采用在凝胶固化样品的表面覆上纳米银颗粒来进一步研究提高LIBS光谱强度的方法。强酸性水样中的 Cu2+、Ni2+和Zn2+ 浓度仍为100 mg∙L−1,硫酸浓度为1.0×105 mg∙L−1,固化剂(等物质的量的丙烯酰胺和丙烯酸)浓度为5 mol∙L−1。对样品进行紫外光照凝胶固化后,用LIBS分析方法分别检测了表面覆有纳米银颗粒和无纳米银颗粒的凝胶固体样品。LIBS分析时的激光能量为150 mJ,数据采集的延迟时间为155 ns。对LIBS分析得到的光谱进行定性分析,得到Cu(I)、Ni(I) 和Zn(II)的特征谱线分别为324.75、218.64和206.12 nm。同时,凝胶固化样品表面覆有纳米银颗粒后,LIBS激发的Cu(I) 324.75 nm、Ni(I) 218.64 nm 和Zn(II) 206.12 nm光谱强度与无纳米银颗粒情况相比分别提高了5.56、19.31和8.73倍(见图5)。样品表面的显微图像(见图 6)分析表明,凝胶样品表面的纳米银颗粒能极大地提高激光对样品的烧蚀程度,从而增强LIBS光谱的强度。
分析其作用机理发现:由于样品表面的纳米颗粒能有效提高LIBS强度,激光脉冲与样品表面的纳米银颗粒发生反应,并诱导纳米银颗粒中传导电子的干涉和集体振荡,从而放大了激发产生的磁场;同样纳米颗粒表面附近和纳米颗粒间隙中的电场也增加了,局部电场的增强导致入射激光束的局部强度增加几个数量级,使得样品表面经击穿产生的等离子体中的电子数增加,最终使等离子发射的光谱强度增强[16-17]。
-
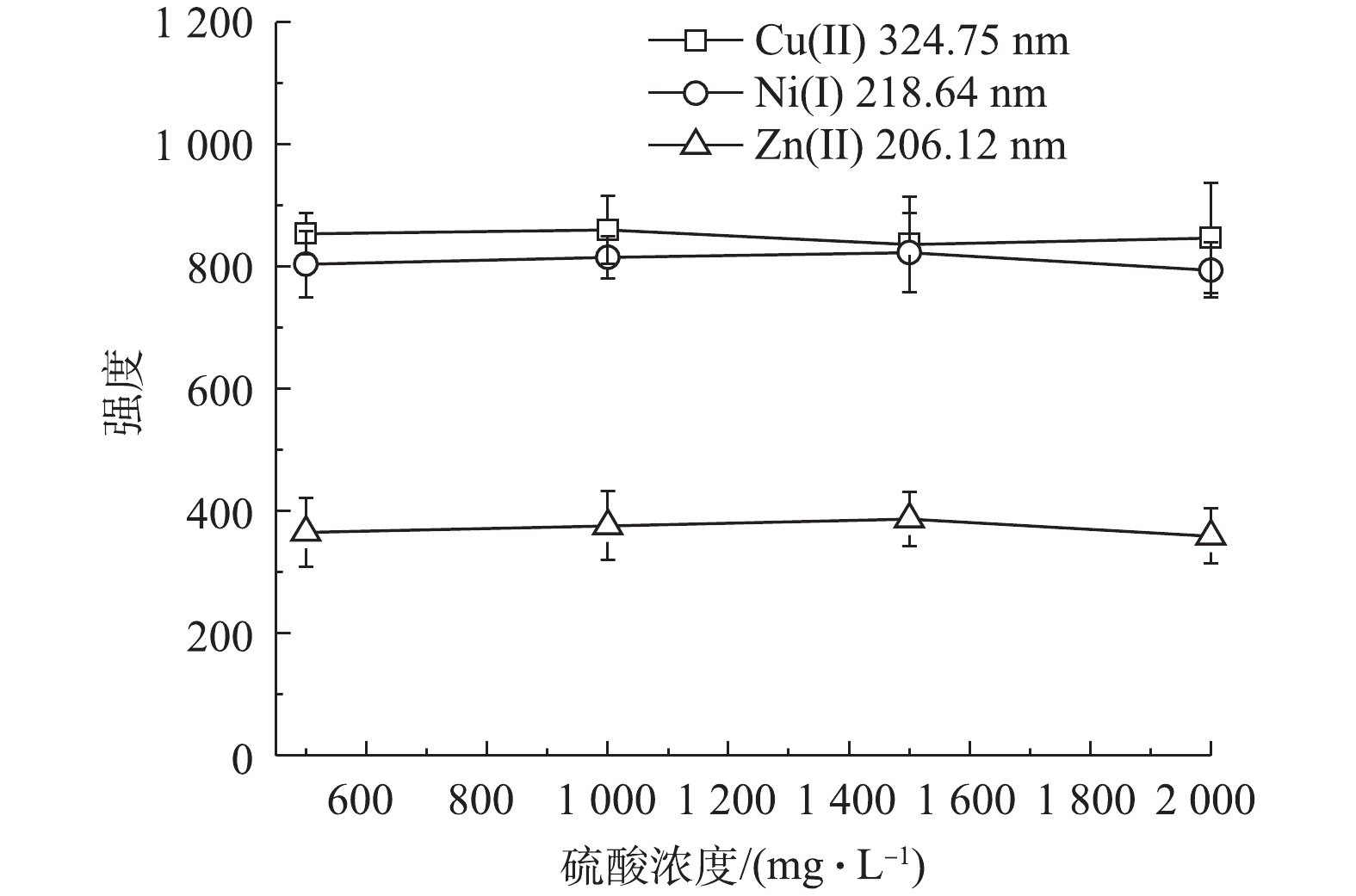
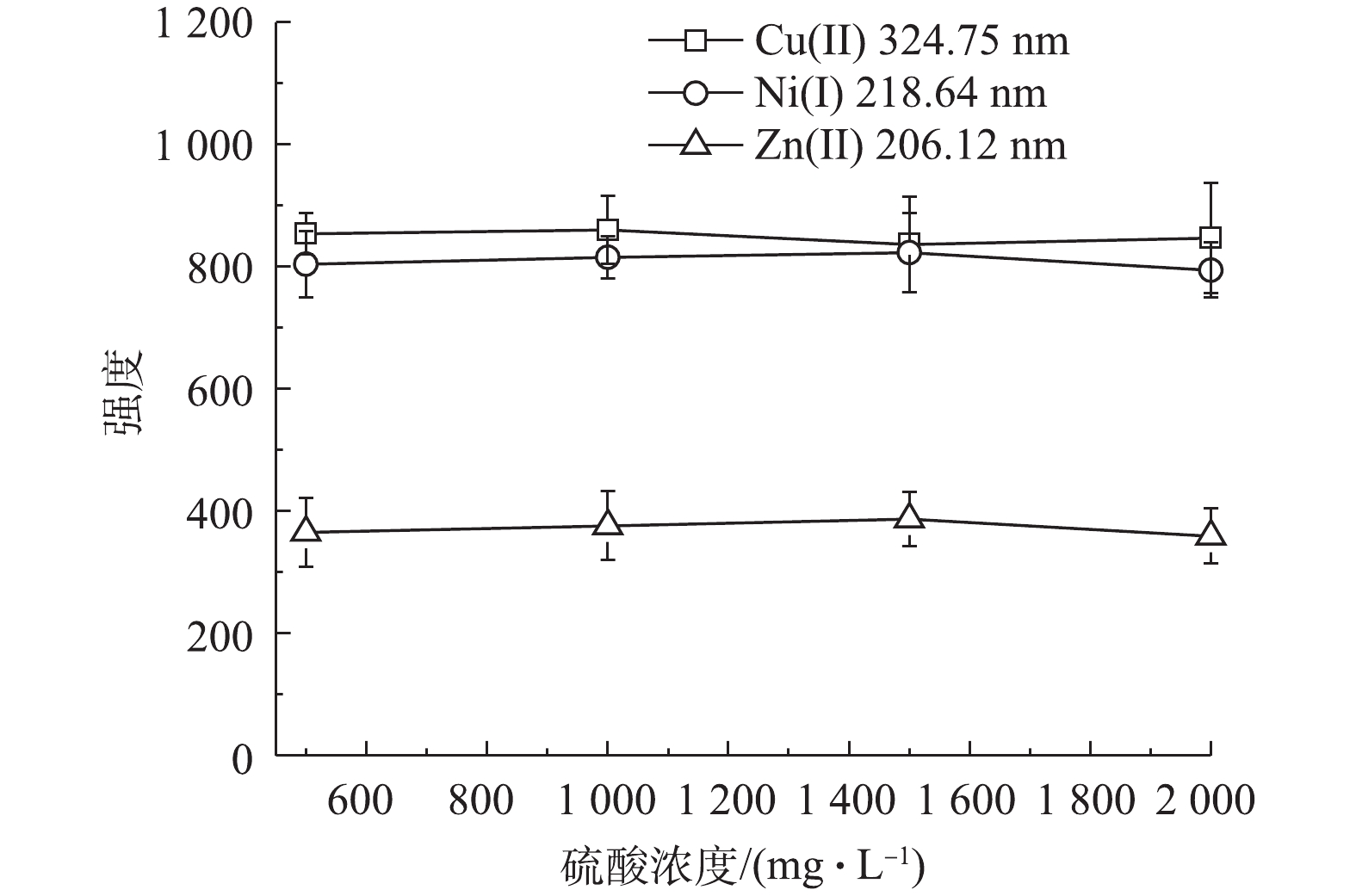
酸浓度对分析结果的影响也较大,本研究继续考察了水样中硫酸浓度对样品凝胶固化和纳米银颗粒增强LIBS的影响。配制水样中Cu2+、Ni2+和Zn2+浓度均为100 mg∙L−1,硫酸浓度分别为0.5×105、1.0×105、1.5×105和2×105 mg∙L−1。在经过紫外光照凝胶固化和覆上纳米银颗粒后,在激光强度为150 mJ,延迟时间为155 ns的条件下,对样品进行LIBS分析,得到Cu2+、Ni2+和Zn2+的特征光谱强度。结果表明,随着水样中硫酸浓度的增加,样品经激光激发得到的3种重金属特征光谱的强度均没有明显变化(见图7),即水样中酸浓度不会影响固化样品经LIBS分析得到的光谱强度。这也说明该方法适用于LIBS分析前对不同酸浓度废水的固化处理。
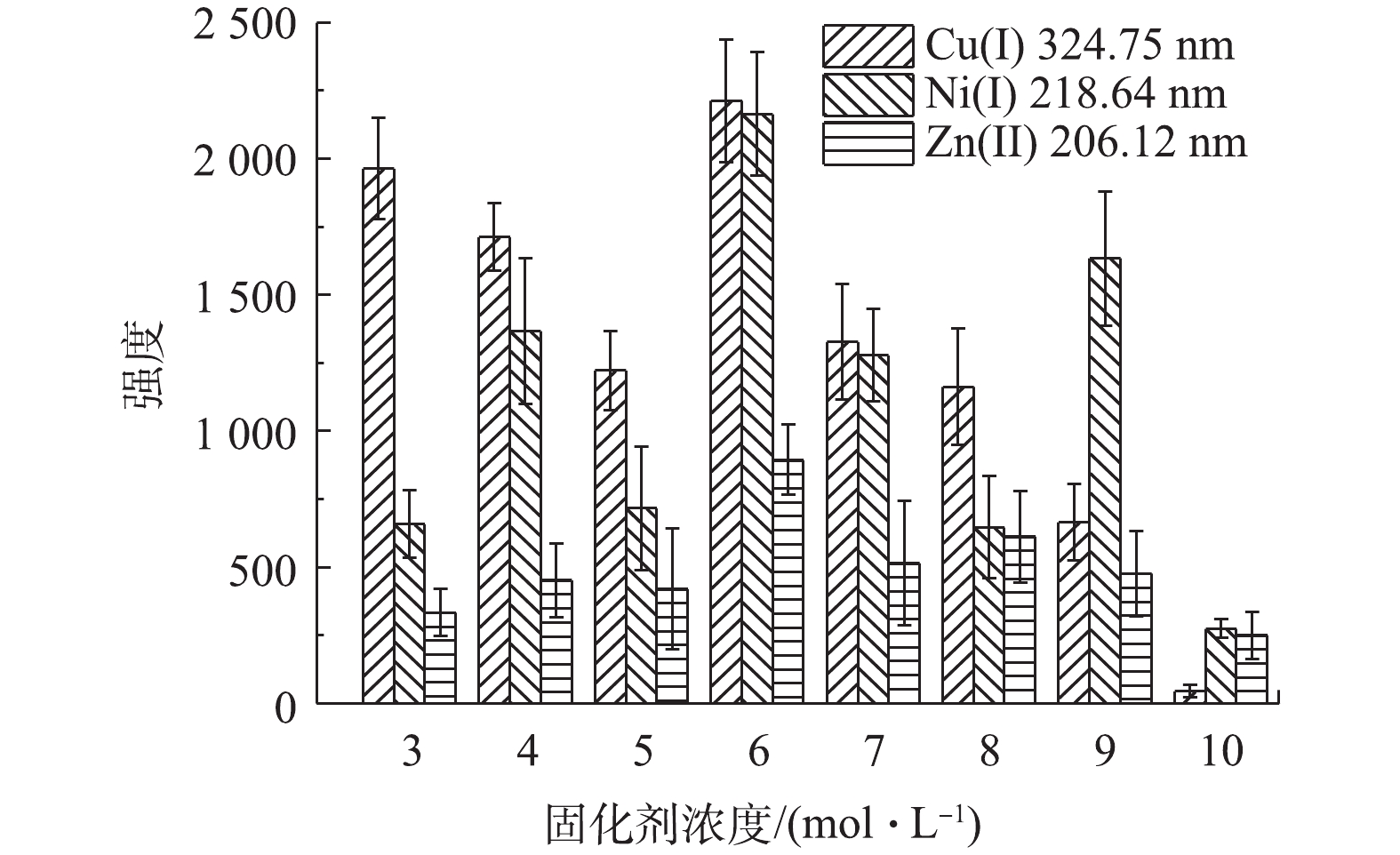
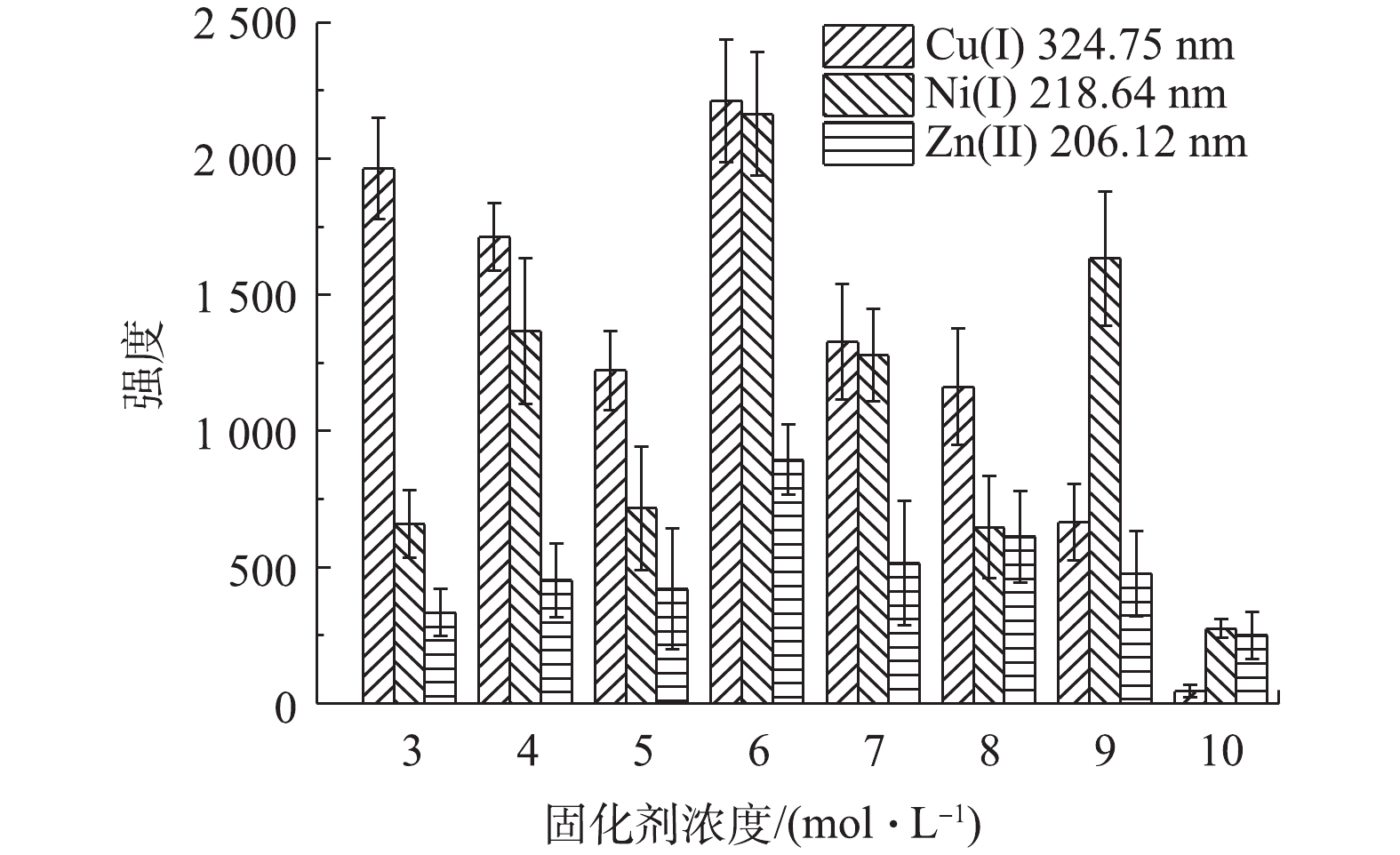
进一步研究固化剂浓度对固化效果和固化后凝胶样品中重金属含量的影响,配制重金属浓度均为100 mg∙L−1,硫酸浓度为1.0×105 mg∙L−1的水样,在固化剂(等物质的量的丙烯酰胺和丙烯酸)浓度分别为2、3、4、5、6、7、8、9和10 mol∙L−1的条件下,按照图2流程进行固化。结果表明:浓度为2 mol∙L−1时,样品不能固化;经LIBS分析得到在固化剂浓度为6 mol∙L−1时,Cu(I) 324.75 nm、Ni(I) 218.64 nm和Zn(II) 206.12 nm特征光谱强度均最强(见图8)。因此,可确定丙烯酰胺和丙烯酸的最佳浓度为6 mol∙L−1。
-
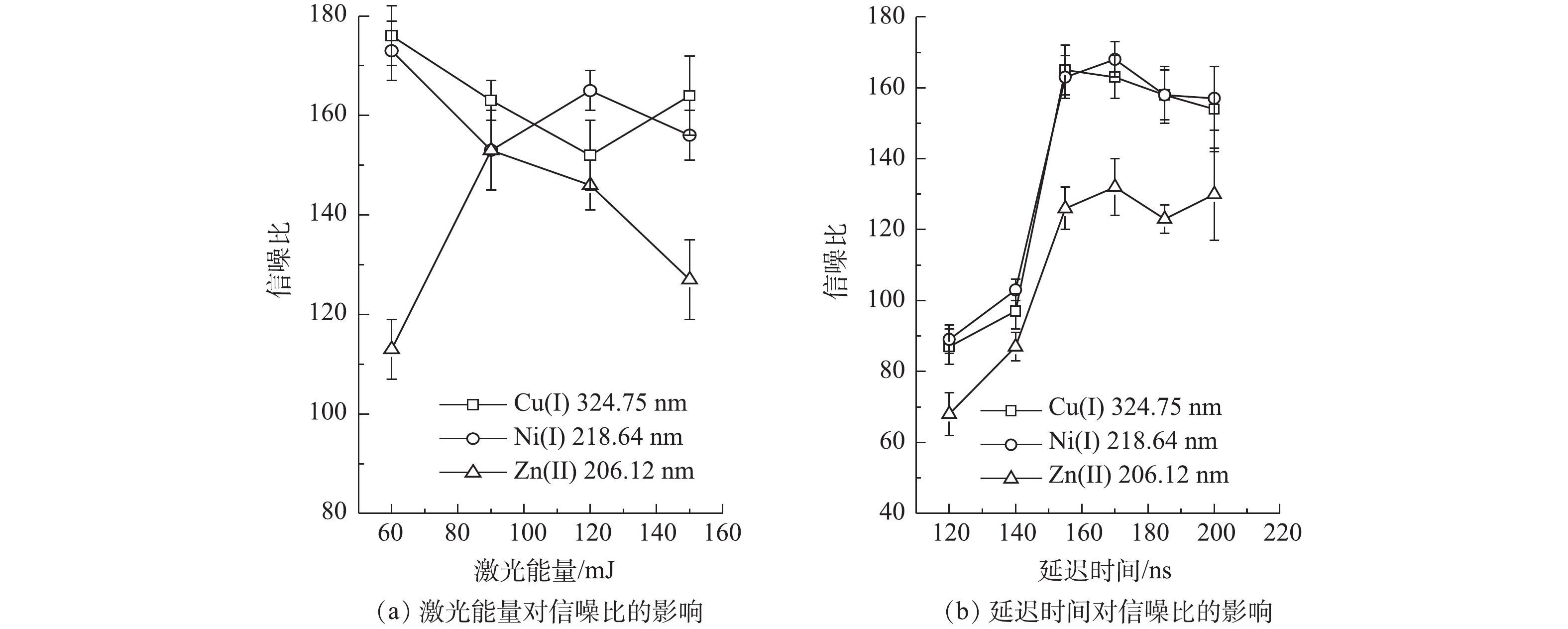
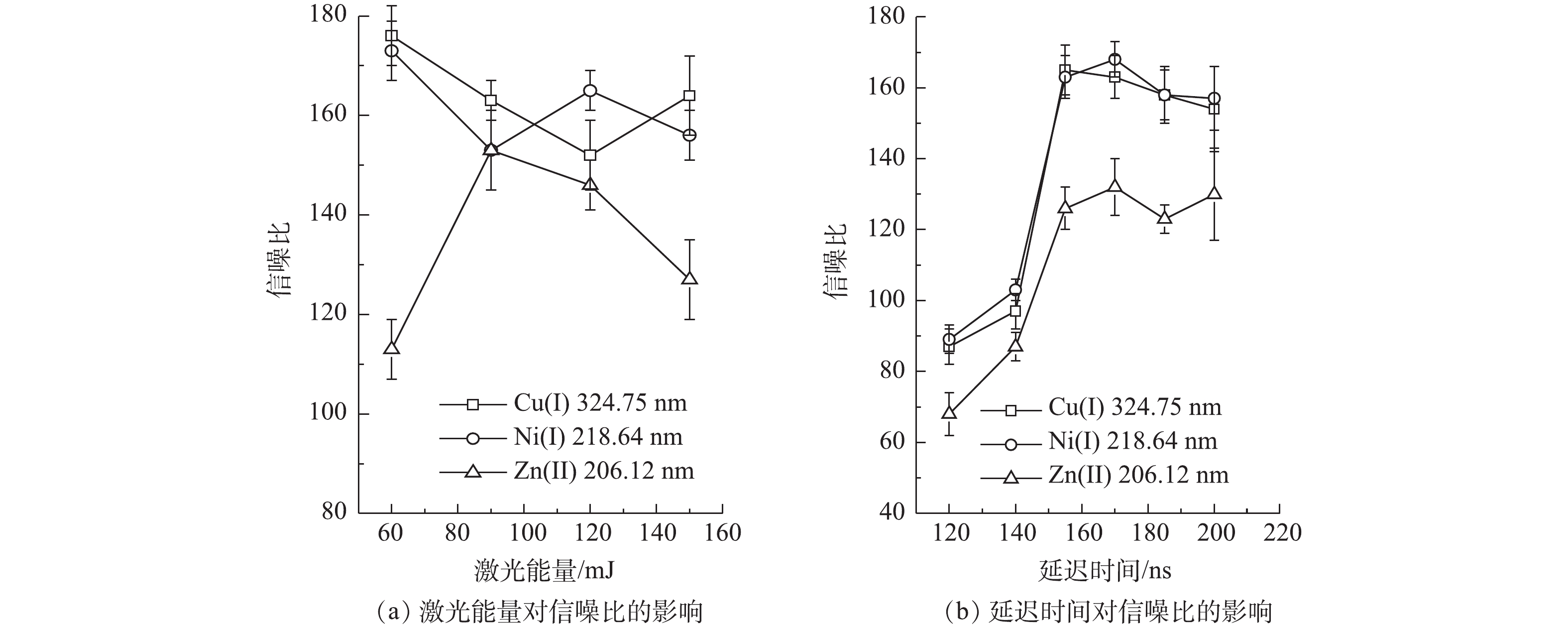
激光能量和光谱仪数据采集的延迟时间是影响LIBS检测强度的2个重要因素。为得到更好的LIBS检测效果,对LIBS系统中的激光强度和数据采集延迟时间进行了优化。选择经过紫外光照凝胶固化,并覆有纳米银颗粒的样品,在不同的激光强度下进行激光击穿,得到Cu2+、Ni2+和Zn2+的特征光谱强度,通过计算得出信噪比。结果表明:同时分析重金属Cu2+、Ni2+和Zn2+时,最佳的激光能量为120 mJ(见图9(a));在激光能量为120 mJ的条件下,进一步对采集数据的延迟时间进行优化,优化范围为120~200 ns;当延迟时间为155 ns时,LIBS检测Cu2+、Ni2+和Zn2+这3种重金属的信噪比最高(见图9(b))。因此,确定155 ns为光谱仪的最佳数据采集延迟时间。
-
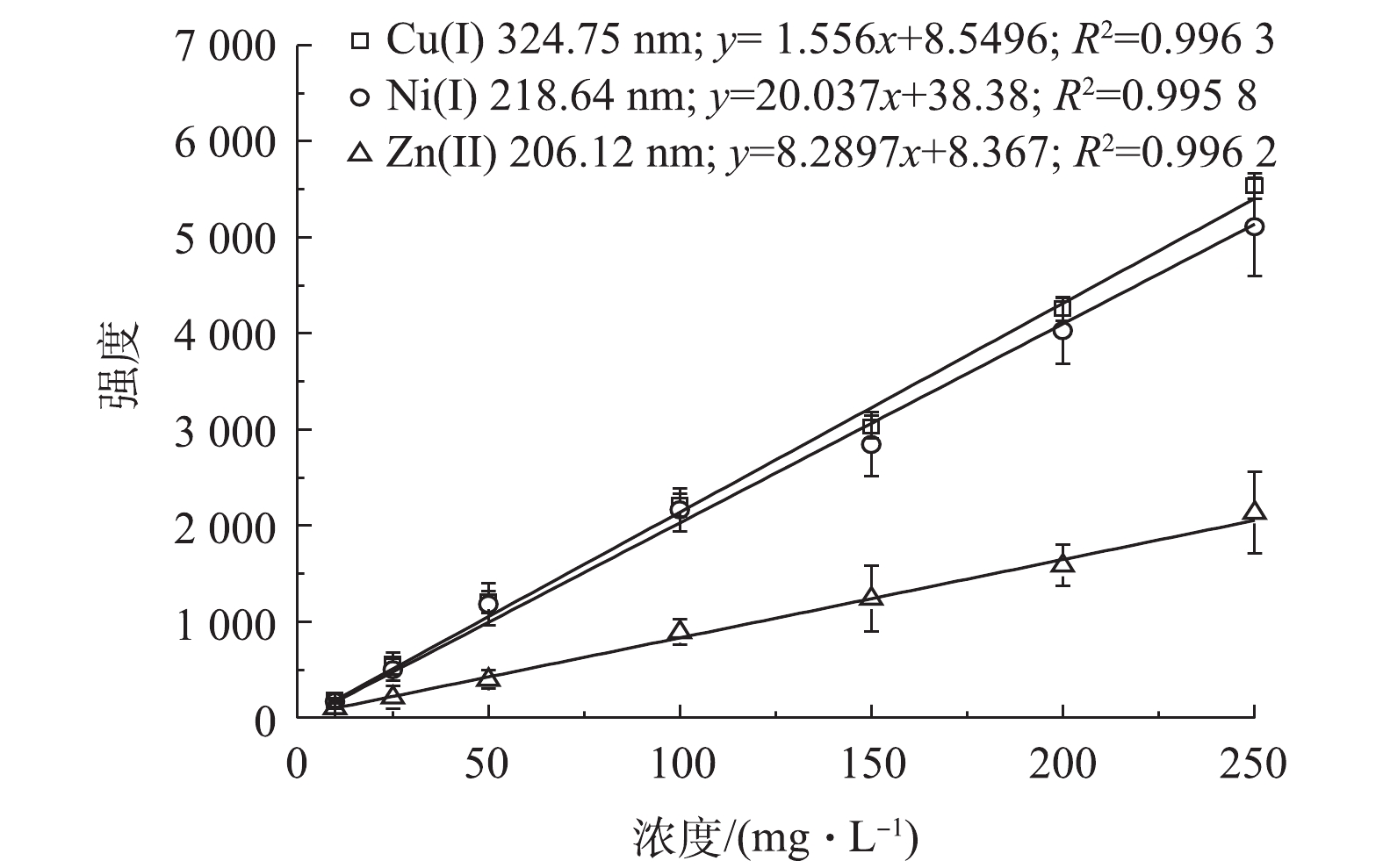
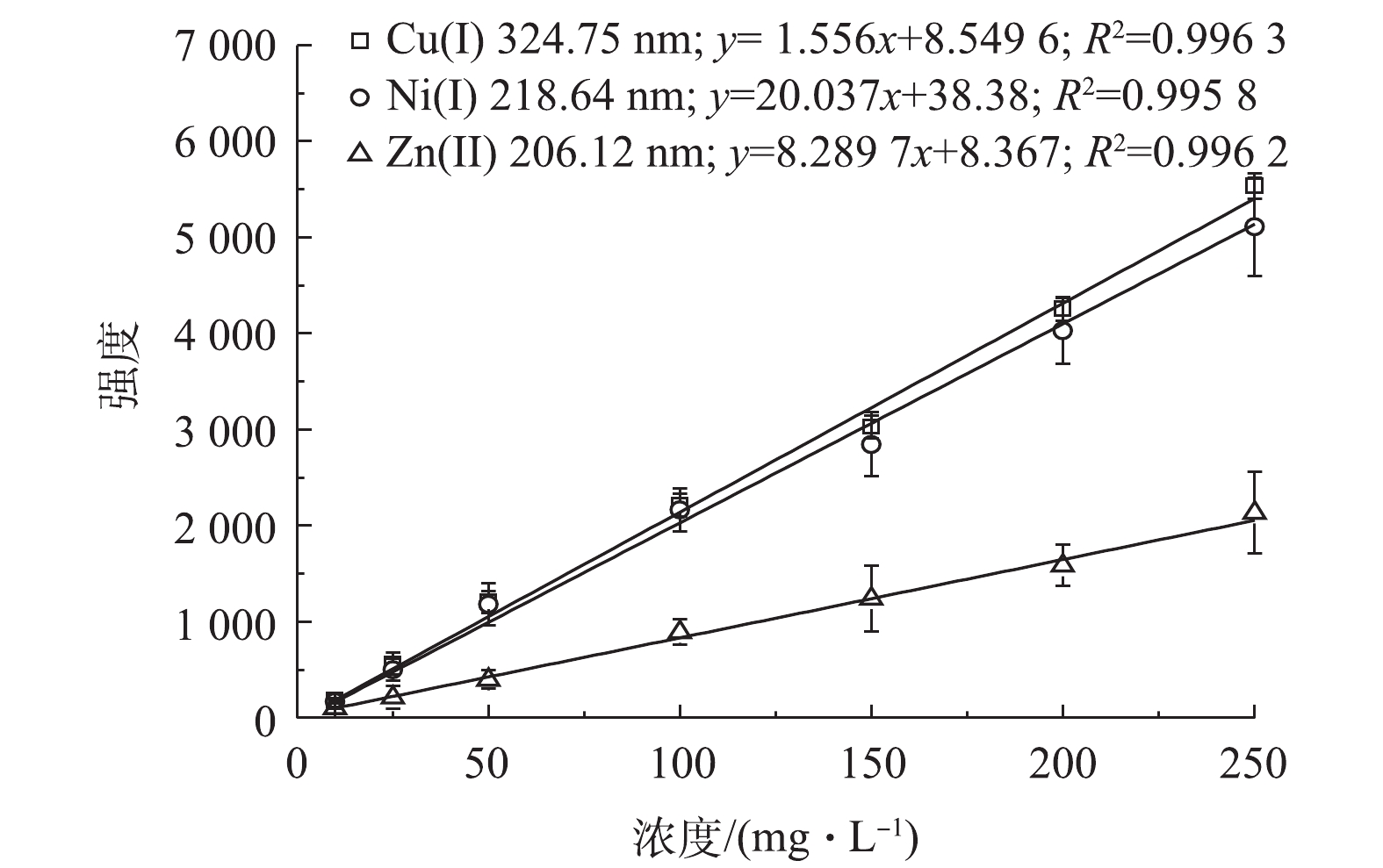
配制硫酸浓度为1.0×105 mg∙L−1,以及Cu2+、Zn2+和Ni2+的浓度分别为10、25、50、100、150、200和250 mg∙L−1的一系列标准溶液。在最优样品固化条件下,按照图2所示流程进行固化处理,在最佳条件下进行LIBS分析,得出Cu、Zn和Ni 3种重金属的光谱强度;再对不同浓度下3种重金属的LIBS光谱强度进行线性拟合,从而得到其LIBS分析标准曲线。结果表明,Cu、Ni和Zn特征谱线强度在0~250 mg∙L−1浓度范围内,具有良好的线性关系,拟合度分别为0.996 3、0.995 8和0.996 2(见图10)。说明采用紫外光照凝胶固化-纳米颗粒增强LIBS分析强酸性废水样品时,所得到的重金属特征谱线的光谱强度与液体样品中重金属的浓度具有良好的线性关系。
检出限(limit of detection,LOD)是分析方法中非常重要的参数,表示分析方法对待测元素的最低检测浓度,检出限浓度的计算公式[13]见式(1)。
式中:CL为检出限浓度;σ为背景信号的标准偏差;S为标准曲线的斜率。
本实验对空白样品进行50 次 LIBS 光谱数据采集。背景信号的标准偏差(σ)由空白样品经LIBS采集得到的光谱数据计算得到;斜率(S)由所得的标准曲线确定;定量分析校准曲线的主要参数值列于表1。根据表1中σ和S的数值,按照式(1)计算出 Cu2+、Ni2+和Zn2+ 的检出限浓度(CL)分别为 1.489、1.512和 4.886 mg∙L−1。
-
实际强酸废水采集自某有色金属冶炼企业,其中含有的硫酸、Zn2+、Cu2+、F−、Cl−、Al3+、Pb2+和Fe2+的浓度分别为2.04×105、67.77、8.77、1 644.00、10 400.00、17.27、5.61和59.42 mg∙L−1。在实际强酸废水中添加已知浓度的Cu2+、Ni2+和Zn2+,然后按照优化后的固化条件对样品进行处理,在最佳LIBS条件下进行检测,得到水样中Cu2+、Ni2+和Zn2+的浓度,并根据式(2)计算回收率,用以评价该方法对实际废水中重金属检测的准确性,结果如表2所示。
式中:R为相对标准偏差;C测定为实际测定重金属浓度,mg∙L−1;C初始为实际废水中重金属浓度,mg∙L−1;C添加为添加的重金属浓度,mg∙L−1。
由表2可知,采用紫外光照凝胶固化-纳米颗粒增强LIBS对强酸废水中Cu2+、Ni2+和Zn2+分析的加标回收率分别为109.00%、91.20%和92.50%。说明此方法对强酸废水中重金属的浓度分析是接近真实浓度的。另外,表2显示3种重金属元素分析的相对标准偏差(RSD)分别为12.21%、10.05%和11.15%,说明该方法分析所得的数据稳定性良好。
2.1. 固化效果及机理
2.2. AgNPs对LIBS的增强效果及机理
2.3. 固化条件优化
2.4. LIBS分析方法的条件优化
2.5. 标准曲线和检出限
2.6. 实际样品分析和回收率
-
1)在紫外光照条件下,强酸性水样中丙烯酰胺和丙烯酸在光引发剂(2-羟基-4′-(2-羟乙氧基)-2-甲基苯丙酮)作用下聚合,并通过氢键作用形成相互交联的长链聚合物网络结构,可在广泛的酸度条件下实现对强酸性废水的高效凝胶固化。
2)凝胶固化样品表面覆上纳米银颗粒能明显提高LIBS对样品的烧蚀程度,从而提高激发光谱强度。与无纳米银颗粒情况相比,纳米银颗粒显著增强了LIBS激发产生的Cu(I) 324.75 nm、Ni(I) 218.64 nm 和Zn(II) 206.12 nm特征光谱强度。
3)在优化的固化条件和LIBS系统参数条件下,紫外光照凝胶固化-纳米颗粒增强LIBS 检测强酸性水样中Cu2+、Ni2+ 和Zn2+ 的标准曲线线性良好,检出限分别为1.489、1.512和 4.886 mg∙L−1。实际强酸性废水的加标回收测定结果表明该方法对强酸性废水中重金属分析具有良好的准确性和稳定性。
4)紫外光照凝胶固化-纳米颗粒增强LIBS可适用于复杂强酸性废水中重金属元素的快速、准确的定性和定量分析,为强酸性废水中重金属的快速检测提供新的技术支持。





 下载:
下载: